More than a century ago, a Russian zoologist peering through a microscope at transparent starfish larvae laid the foundations of modern immunology. In the 1880s, Ilya Ilyich Metchnikoff observed mobile cells rushing to surround tiny citrus thorns he had inserted into the larvae’s bodies. From these simple experiments a radical idea emerged: that certain white blood cells actively engulf and destroy harmful agents — a process he named phagocytosis.
At the time, Metchnikoff’s proposal that inflammation was protective rather than pathological was met with skepticism from leading figures such as Louis Pasteur and Emil von Behring. Yet history would vindicate him. Metchnikoff was awarded the Nobel Prize in 1908 and today phagocytosis is a central element of immunology.
Now, more than a hundred years later, new work led by GIMM’s researchers, Rui Martins and Birte Blankenhaus, from Miguel Soares Lab, has expanded Metchnikoff’s original vision. Their study, published at The EMBO Journal (Homeostatic control of energy metabolism by monocyte-derived macrophages | The EMBO Journal | Springer Nature Link), reveals that macrophages do far more than defend the body against infection. They act as metabolic guardians, capable of sensing systemic stress, recycling damaged cellular components and sustaining the function of entire organs when fundamental metabolic processes fail, keeping tissues functioning, such as the heart beating when otherwise mice would die following disruption of iron homeostasis.
Ten years to uncover an invisible rescue system
The findings are the result of nearly a decade of research, dozens of animal models and an extensive series of technically demanding experiments. The outcome is a 45-page study, now published in The EMBO Journal, that uncovers a previously unrecognised mechanism linking iron metabolism, mitochondrial health and whole-body energy balance.
“This was a very long project, with many experimental layers, because we wanted to be absolutely certain that what we were seeing was real and robust,” explains Rui Martins, first author of the study. “Bone marrow transplants [a procedure done throughout the work] alone take months to complete and have to be performed in parallel across multiple models.”
Multicellular life depends on constant communication between organs to maintain vital parameters such as energy production, body temperature and nutrient balance. When this network is disrupted, the consequences are often catastrophic. The new study shows that when iron metabolism collapses, macrophages step in as central coordinators of systemic rescue.
Iron: essential, reactive and indispensable
Iron lies at the heart of this story. It is an essential cofactor for numerous metabolic reactions, particularly in the mitochondria, the energy factory of the cell, where it supports the electron transport chain and the production of the energy molecule, ATP.
“Iron is involved in an extraordinary number of metabolic processes,” says Rui Martins. “But it’s also extremely reactive. If it were free inside the cell, it would catalyse highly damaging oxidative reactions.”
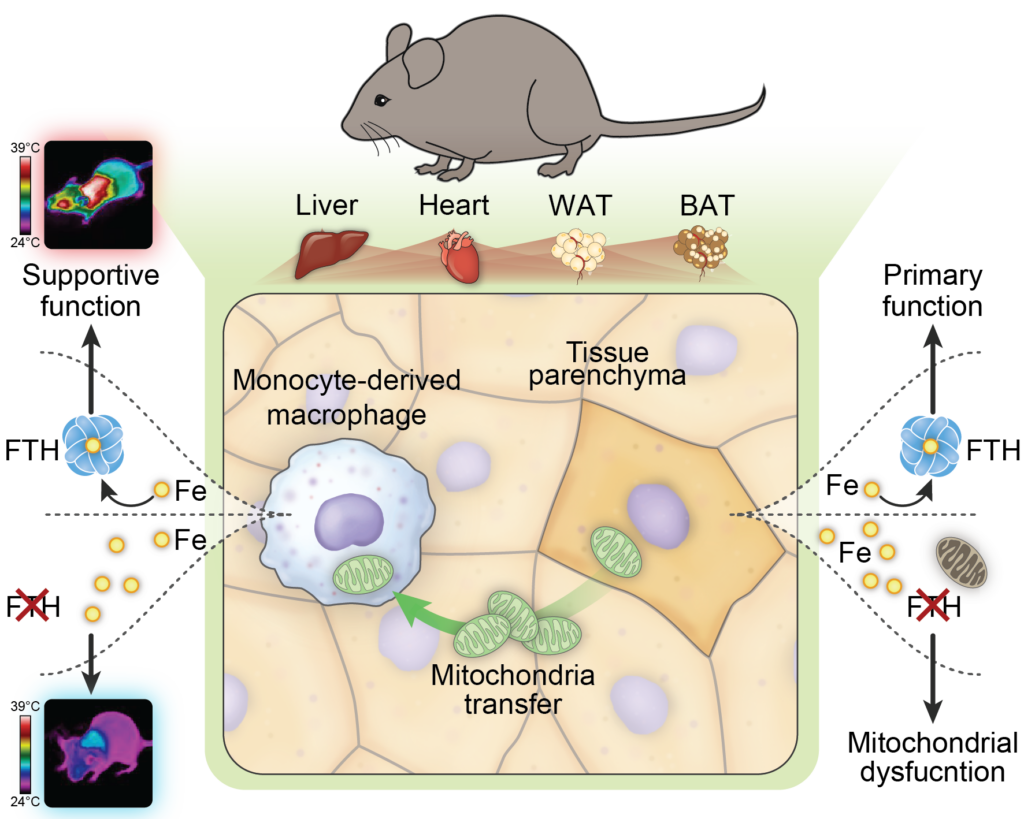
To manage this risk, cells tightly control iron using ferritin, a protein complex that safely stores the metal. In this study, researchers genetically deleted the heavy chain of ferritin (FTH) in mouse tissues, creating a controlled disruption of iron storage across the organism.
The result, as previously published, was a profound metabolic collapse. Despite iron being present, cells were unable to use it safely or efficiently — a state the researchers describe as a “false iron deficiency”. Animals developed dysfunctional mitochondria, atrophied adipose tissue, cardiac abnormalities, impaired thermogenesis and ultimately multi-organ failure and death.
After that, the researchers transplanted bone marrow cells that express FTH into recipient mice that do not express FTH and surprisingly, these mice survived. Conversely, when they instead transplanted bone marrow cells, where only the macrophage lineage does not express FTH, the mice could no longer be rescued, directly implicating macrophages in the rescue from metabolic collapse and death.
“This tells us that even though ferritin is essential for survival, it’s sufficient for macrophages to express it for the whole organism to function,” Rui Martins explains. “Tissues can be completely unable to make ferritin, and the animal still survives.”
The study reinforces the notion that monocyte-derived macrophages act as sensors of iron imbalance, detecting metabolic distress in tissues and launching a coordinated response that prevents organ failure. The researchers describe this role as that of a cellular “ferrostat” — a regulator that links iron handling to energy metabolism at the organismal level.
But how do macrophages achieve this?
Transmitophagy: mitochondrial waste management at scale
In tissues lacking ferritin, mitochondria accumulate oxidative damage and become dysfunctional. Normally, cells would remove this waste through a cleaning process called mitophagy, which is an energy-demanding process. But under severe metabolic stress, this capacity collapses.
What the study shows is that stressed cells export their damaged mitochondria to macrophages. The macrophages then engulf and degrade them in lysosomal compartments — a process referred to as “transmitophagy”.
“It’s almost like an organism-wide waste management system,” Martins explains. “Cells outsource the clearance of damaged mitochondria to macrophages, which are better equipped to deal with the iron and oxidative burden.”
Notably, this finding overturns the team’s initial hypothesis. “We thought macrophages might be donating healthy mitochondria to tissues,” he says. “The data showed the opposite — the flow is mainly from stressed cells to macrophages.”
Handling this mitochondrial influx requires macrophages themselves to be metabolically robust. The study shows that FTH expression activates a transcriptional programme driven by TFAM (Mitochondrial Transcription Factor A), a key regulator of mitochondrial DNA maintenance and biogenesis.
When TFAM was deleted specifically in macrophages, the rescue effect vanished — even though ferritin was still present. The result demonstrates that macrophage mitochondrial health is essential for sustaining whole-body metabolic rescue.
Beyond iron: implications for disease and physiology
Although the experimental model relies on an artificial disruption of iron metabolism, the authors believe it reveals a much broader biological principle. Similar macrophage-mediated mitochondrial clearance has already been reported in the heart, where its failure leads to cardiac disease even without iron defects.
This versatility also raises difficult questions. In cancer, for example, tumour cells experience intense metabolic stress. “What stops a cancer cell from asking a macrophage for help?” Martins asks. By supporting stressed tissues, macrophages may — in some contexts — inadvertently promote disease.
Seen in historical perspective, the findings represent a direct conceptual extension of Metchnikoff’s original insight. Phagocytosis is no longer just a defence against microbes; it is part of a sophisticated system of cellular maintenance, metabolic support and physiological resilience.
“This work shows that macrophages sit at the intersection of immunity, metabolism and organ communication,” Martins concludes. “They really are the Swiss army knives of biology — adaptable, versatile and indispensable for life.”